Supplier:
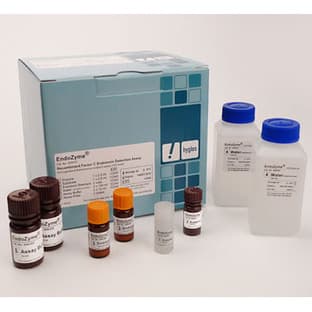
Hyglos GmbHEndoZyme® - Endotoxin Detection Assay
The endotoxin-specificity of EndoZyme® eliminates all false-positive and elevated results seen with LAL due to ß-glucan reactions in the LAL cascade. Adding the ease-of-use and excellent lot-to-lot consistency, EndoZyme® is a representative of modern recombinant methods, moving away from the animal source which for decades has been the horseshoe crab.
Having a homogeneous assay format without a solid phase, EndoZyme® is best suited for non-complex samples and buffer conditions. For complex sample matrices, EndoLISA® is recommended.
Prices direct from Hyglos GmbH
Quick response times
Exclusive Biosave savings/discounts
SPECIFICATIONS
No specifications available
Please contact the supplier for more information
SUPPLIER INFO
Email:
inquiry@hyglos.deMore from Hyglos GmbH
Latest promotions
Spend less time on DNA cleanup so you can do more science. The MSB Spin PCRapace is the fastest way to purify your DNA from PCR, restriction digestion, and...
New brilliant antibodies, and new lower prices!For flow cytometry reagents in general, \"bright is better.\" The violet-excitable BD Horizon™ BV421 and...
As an incentive to qualify our BSA, we are offering a 20% discount when you purchase your first 100g, 500g or 1000g of any grade of Bovine Serum Albumin....
It is not every day that you are given something for nothing. We are giving away additional spectrophotometer software.Cecil Instruments have enhanced the...
We're so sure that you'll prefer Cayman Assay kits over your present brand that we're willing to give you a free assay kit to prove it!
For the past decade scientists have extensively used ATS secondary toxin conjugates to make their own targeted toxins for in vitro use.The ability to combine...
10% Discount on 2 Rabbit Polyclonal Antibody Service. With over 20 years experience, SDIX has developed into the premier US custom antibody producer,...
Did your supplier increase the price of Fetal Bovine Serum? Did they substitute the US Origin with USDA? Well say no more! Innovative Research is still...
Bulk Cytokines with Custom Vialing.20 - 50% off cytokines, growth factors, chemokines and more...For a limited time Cell Sciences is offering substantial...
Are you planning to have a customised antibody made for your research?Since 2000, Everest has been producing a catalog containing thousands of affinity...
Jenway’s 73 series spectrophotometer range provides four models with a narrow spectral bandwidth of 5nm and an absorbance range of –0.3 to 2.5A,...
Top suppliers
United States Biological
230747 products
Carl Zeiss Microscopy
27 products
Promega Corporation
11 products
Panasonic Healthcare Company
5 products
Life Technologies
1 products
Nikon Instruments Europe
11 products
Olympus Europa Holding GmbH
3 products
Leica Microsystems, Inc.
10 products
GE Healthcare Life Sciences
2 products
Tecan Trading AG
19 products
Beckman Coulter, Inc.
1 products
AB SCIEX
3 products
BD (Becton, Dickinson and Company)
1 products
RANDOX TOXICOLOGY
5 products
Randox Food Diagnostics
6 products